1. Fundamental Concepts
- Definition: Lewis Electron Dot Structures (LEDS) are diagrams used to show how valence electrons are arranged among atoms in a molecule or ion. They help understand bonding patterns, molecular shape, and formal charges.
- Valence Electrons: The outermost electrons of an atom, which are involved in chemical bonding.
- Octet Rule: Atoms tend to gain, lose, or share electrons to achieve a full valence shell of eight electrons, except for hydrogen and helium, which aim for two electrons.
2. Key Concepts
Steps to Draw LEDS:
- Sum Valence Electrons: Count the total valence electrons for all atoms. Add electrons for negative charges and subtract for positive charges.
- Determine central atom: Usually the least electronegative, except H.
- Skeleton Structure: Connect the central atom to surrounding atoms with single bonds (2 electrons each).
- Complete Octets: Distribute remaining electrons to the surrounding atoms (terminal atoms) to satisfy the octet rule.
- Assign Remaining Electrons: Place any leftover electrons on the central atom.
- Form Multiple Bonds: If the central atom lacks an octet, move lone pairs from terminal atoms to form double or triple bonds.
3. Examples
Example 1 (Easy)
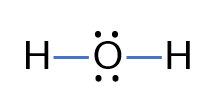
Problem: Draw the Lewis Electron Dot Structure for water (H₂O).
Step-by-Step Solution:
- Total valence electrons:
- Central atom: Oxygen (O), surrounded by Hydrogen (H)
- Draw single bonds between O and H:
- Complete the octets: Each H has 2 electrons, O has 6 electrons (2 from each H and 4 lone pairs).
- Final structure:
Example 2 (Medium)
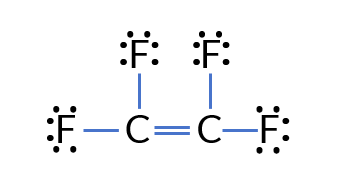
Problem: Draw the Lewis Electron Dot Structure for C₂F₄.
Step-by-Step Solution:
- Total valence electrons: 4 × 2 + 7 × 4 = 36
- Central atom: Carbon (C), surrounded by fluorine (F)
- Draw single bonds between C and O: C−C
- Complete the octets: Each F needs 1 more electron, and form a C=C double bond so each carbon reaches an octet.
- Final structure:
Example 3 (Hard)
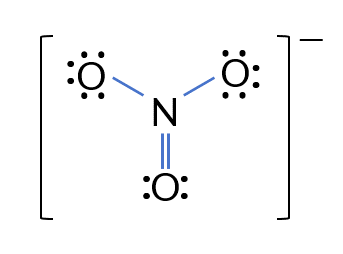
Problem: Draw the Lewis Electron Dot Structure for the nitrate ion NO₃⁻.
Step-by-Step Solution:
- Total valence electrons: 5 + 6 × 3 + 1 = 24
- Central atom: Nitrogen (N), surrounded by Oxygen (O)
- Connect N to three O atoms with single bonds.
- Complete the octets: Nitrogen initially has only 6 electrons → form one N=O double bond.
- Final structure:
4. Problem-Solving Techniques
- Visual Strategy: Use a step-by-step approach to draw the structure, starting with the central atom and adding surrounding atoms and bonds.
- Recognize Common Patterns (e.g., carbon forms 4 bonds, nitrogen forms 3 bonds + 1 lone pair, etc.)
- Connect Lewis Structures to later topics: Lewis structures → VSEPR → molecular shape → polarity → intermolecular forces