1. Fundamental Concepts
Definition: The rate law is a mathematical equation that links the rate of a chemical reaction to the concentration of the reactants.
General Mathematical Form:
For a reaction $A + B \rightarrow Products$, the rate law is expressed as:
$$Rate = k[A]^x [B]^y$$
Key Components:
Rate: The speed of the reaction (usually in M/s).
$k$ (Rate Constant): A specific constant for the reaction at a given temperature. It does not change with concentration.
$[A], [B]$: Molar concentrations (mol/L) of the reactants.
$x, y$ (Exponents): Numbers that describe how sensitive the rate is to changes in [A] or [B]. Crucial Note: These exponents are NOT the coefficients from the balanced chemical equation. They must be found by experiment.
2. Key Concepts
Concentration Dependence: The rate of a reaction is almost always dependent on the concentration of the reactants. As reactants are used up (concentration decreases), the rate usually slows down.
Isolation of Variables: To find out how [A] affects the rate, you must keep [B] constant and vice versa.
Initial Rate: Chemists usually measure the "Initial Rate" (the speed at the very start of the reaction) because at that moment, the reverse reaction is negligible and the concentrations are known exactly.
3. Examples
Easy
Question: If the concentration of reactant A is doubled, and the rate of the reaction exactly doubles as a result, what is the exponent ($x$) for A in the rate law?
Answer: The exponent is 1.
Reasoning: $2^1 = 2$. (If it quadrupled, the exponent would be 2; if it stayed the same, the exponent would be 0).
Medium
Question: A reaction has the rate law: $Rate = k[X]$. If the initial concentration of X is 0.1 M, the rate is 0.05 M/s. What will the rate be if [X] is increased to 0.3 M?
Answer: The rate will be 0.15 M/s.
Reasoning: The concentration tripled ($0.3 / 0.1 = 3$). Since the exponent is 1, the rate also triples ($0.05 \times 3 = 0.15$).
Hard
Question: Use the experimental data below to determine the rate law for the reaction $A + B \rightarrow C$.
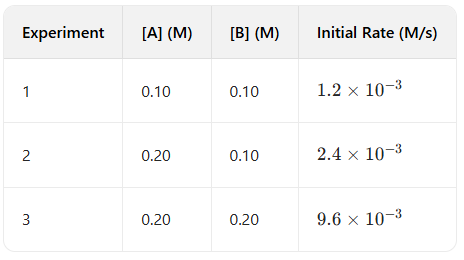
Answer: $Rate = k[A][B]^2$
Analysis:
1. Find exponent for A: Compare Exp 1 and 2. [A] doubles (x2), [B] stays same. Rate doubles (x2). So exponent of A is 1.
2. Find exponent for B: Compare Exp 2 and 3. [B] doubles (x2), [A] stays same. Rate quadruples (x4). Since $2^y = 4$, the exponent of B is 2.
4. Problem-Solving Techniques
The "Ratio Method" :
When solving for exponents, divide the rate of two experiments.
$$ \frac{Rate_2}{Rate_1} = \left(\frac{[Reactant]_2}{[Reactant]_1}\right)^n $$
This cancels out the constant $k$ and allows you to solve for $n$.
Look for the "Control" :
Always scan the data table for two experiments where only one chemical's concentration changed. Ignore the row where both concentrations changed until you know the exponents for the individual chemicals.
Algebraic Solving:
If you know the Rate and the Concentrations, you can plug the numbers back into the equation to solve for $k$ (the Rate Constant).
$$k = \frac{Rate}{[A]^x[B]^y}$$