1. Fundamental Concepts
Definition: Reaction order describes how the rate of a reaction depends on the concentration of the reactants. It is determined experimentally and is not related to the stoichiometric coefficients in the balanced equation.
Rate Law Equation:
: Rate constant (temperature dependent).
: Molar concentrations of reactants.
: Orders with respect to reactants A and B (must be determined by experiment).
Overall Order: The sum of the individual orders ( ).
2. Key Concepts
Zero Order: Rate is independent of concentration ( ). Concentration decreases linearly with time.
First Order: Rate is directly proportional to concentration ( ). Concentration decays exponentially. Half-life ( ) is constant.
Second Order: Rate is proportional to the square of the concentration ( ). Rate is very sensitive to concentration changes.
Units of k:
0th Order:
1st Order:
2nd Order:
Integrated Rate Laws (Linear Plots):
0th: ( vs is linear)
1st: ( vs is linear)
2nd: ( vs is linear)
3. Examples
Easy
Question: Given the rate law , what is the overall order of the reaction?
Answer: 3rd Order.
Explanation: Sum the exponents: .
Medium
Question: A first-order reaction has a rate constant . Calculate the half-life ( ) of the reaction.
Answer: 34.7 seconds.
Explanation: Use the first-order half-life formula:
Hard
Question: Determine the order of the reaction using the following experimental data.
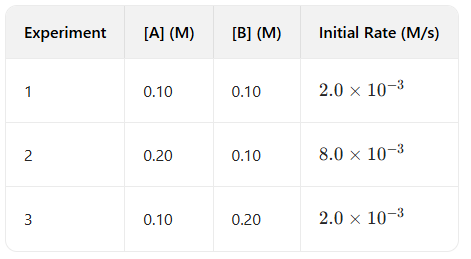
Answer: The reaction is second order with respect to A, zero order with respect to B, and second order overall.
Explanation:
1. Compare Exp 1 and 2: [A] doubles ( ), [B] stays same. Rate quadruples ( ). Since , . (Order in A is 2).
2. Compare Exp 1 and 3: [B] doubles ( ), [A] stays same. Rate stays same ( ). Since , . (Order in B is 0).
3. Overall: .
4. Problem-Solving Techniques
Method of Initial Rates:
Find two experiments where only one reactant's concentration changes.
Use the ratio: .
If Rate when Conc 1st Order.
If Rate when Conc 2nd Order.
If Rate when Conc 0th Order.
Graphical Analysis:
Plot the data. If the y-axis creates a straight line, that defines the order.
Y-axis = 0th Order.
Y-axis = 1st Order.
Y-axis = 2nd Order.
Half-life Trend:
If is constant 1st Order.
If doubles as concentration halves 2nd Order.