1. Fundamental Concepts
- Definition: Resonance structures are different Lewis structures of the same molecule or ion that have the same arrangement of atoms but different arrangements of electrons.
- Formal Charge: A hypothetical charge assigned to an atom in a Lewis structure, assuming that electrons in all chemical bonds are shared equally between atoms, regardless of relative electronegativity. It helps determine the "best" resonance structure.
2. Key Concepts
Formal Charge:
Rules for Drawing Resonance Structures:
Do not move atoms, move only electrons (lone pairs or multiple bonds).
All structures must be valid Lewis structures with correct total valence electrons.
Evaluating Resonance Structures:
1. The most stable structure has the smallest magnitude of formal charges (0 is best).
2. Negative formal charges should reside on the most electronegative atoms.
3. Have a total charge equal to the overall charge on the molecule or ion.
1. The most stable structure has the smallest magnitude of formal charges (0 is best).
2. Negative formal charges should reside on the most electronegative atoms.
3. Have a total charge equal to the overall charge on the molecule or ion.
3. Examples
Example 1 (Easy)
Problem: Draw the resonance structures for the ozone molecule.
Step-by-Step Solution:
- Total Valence Electrons: 6 × 3 = 18 electrons.
- Skeleton: O−O−O.
- Distribute Electrons:
Structure A: Central O has a double bond to the Left O, and a single bond to the Right O.
Structure B: Central O has a single bond to the Left O, and a double bond to the Right O. - Result:
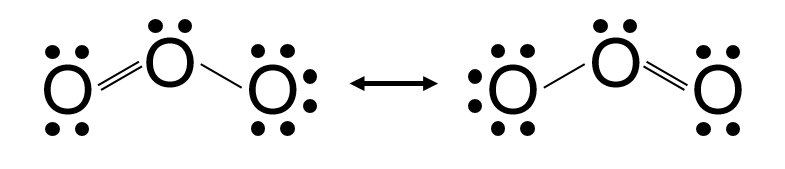
Validation: Each structure has the same number of valence electrons and follows the octet rule.
Example 2 (Medium)
Problem: Determine the formal charges for each atom in one of the resonance structures of .
Step-by-Step Solution:
- Select one of the resonance structures.
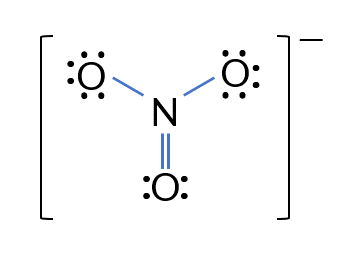
- Calculate the formal charge for each atom using the formula .
For the nitrogen atom:
For the double-bonded oxygen atom:
For the two single-bonded oxygen atoms:
Validation: The sum of the formal charges should equal the overall charge of the ion, which is . Here, , confirming the calculation.
4. Problem-Solving Techniques
- Always calculate formal charge.
- Use formal charge to choose the best structure.
- Remember electronegativity rule: If a negative formal charge is unavoidable, it should be placed on the most electronegative atom.